Page 93 - 《橡塑技术与装备》英文版2026年3期
P. 93
TEST AND ANALYSIS
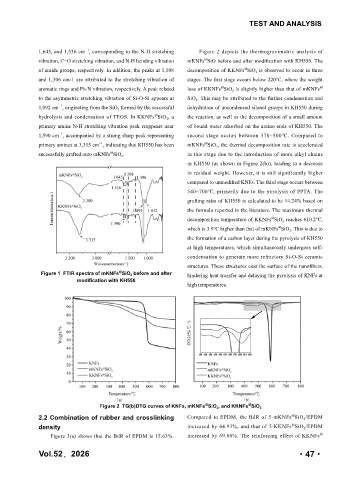
1,645, and 1,536 cm , corresponding to the N-H stretching Figure 2 depicts the thermogravimetric analysis of
-1
vibration, C=O stretching vibration, and N-H bending vibration mKNFs SiO before and after modification with KH550. The
@
@
of amide groups, respectively. In addition, the peaks at 1,508 decomposition of KKNFs SiO 2 is observed to occur in three
and 1,306 cm-1 are attributed to the stretching vibration of stages. The first stage occurs below 220℃, where the weight
@
aromatic rings and Ph-N vibration, respectively. A peak related loss of KKNFs SiO 2 is slightly higher than that of mKNFs @
to the asymmetric stretching vibration of Si-O-Si appears at SiO 2 . This may be attributed to the further condensation and
-1
1,092 cm , originating from the SiO 2 formed by the successful dehydration of uncondensed silanol groups in KH550 during
hydrolysis and condensation of TEOS. In KKNFs SiO 2 , a the reaction, as well as the decomposition of a small amount
@
primary amine N-H stretching vibration peak reappears near of bound water adsorbed on the amino ends of KH550. The
-1
1,590 cm , accompanied by a strong sharp peak representing second stage occurs between 370~500℃. Compared to
primary amines at 3,315 cm , indicating that KH550 has been mKNFs SiO 2 , the thermal decomposition rate is accelerated
-1
@
@
successfully grafted onto mKNFs SiO 2 . in this stage due to the introduction of more alkyl chains
in KH550 (as shown in Figure 2(b)), leading to a decrease
in residual weight. However, it is still significantly higher
compared to unmodified KNFs. The third stage occurs between
540~700℃, primarily due to the pyrolysis of PPTA. The
grafting ratio of KH550 is calculated to be 14.24% based on
the formula reported in the literature. The maximum thermal
decomposition temperature of KKNFs SiO 2 reaches 610.2℃,
@
@
which is 3.9℃ higher than that of mKNFs SiO 2 . This is due to
the formation of a carbon layer during the pyrolysis of KH550
at high temperatures, which simultaneously undergoes self-
condensation to generate more refractory Si-O-Si ceramic
structures. These structures coat the surface of the nanofibers,
@
Figure 1 FTIR spectra of mKNFs SiO 2 before and after hindering heat transfer and delaying the pyrolysis of KNFs at
modification with KH550
high temperatures.
@ @
Figure 2 TG(b)DTG curves of KNFs, mKNFs SiO 2 , and KKNFs SiO 2
2.2 Combination of rubber and crosslinking Compared to EPDM, the BdR of 5-mKNFs SiO 2 /EPDM
@
@
density increased by 64.93%, and that of 5-KKNFs SiO 2 /EPDM
Figure 3(a) shows that the BdR of EPDM is 15.63%. increased by 69.80%. The reinforcing effect of KKNFs @
Vol.52,2026 ·47·