Page 9 - 《橡塑技术与装备》英文版2025年12月
P. 9
SPECIAL AND COMPREHENSIVE REVIEW
increases, and the PET depolymerization effect is
significant. However, if the temperature is set too high,
the reflux of ethylene glycol increases significantly,
energy consumption increases, and the color of the product
gradually deepens. However, after calculation, the yield
of the reaction product PHET does not show a significant
increase.Therefore, the optimal reaction temperature is
controlled at 200 ℃.
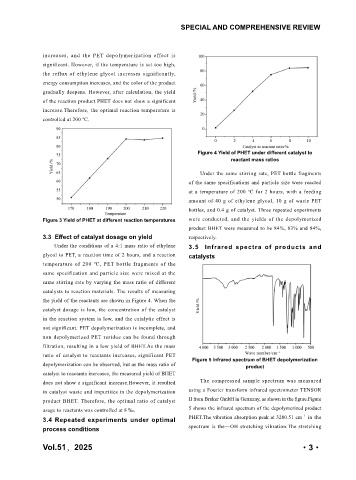
Figure 4 Yield of PHET under different catalyst to
reactant mass ratios
Under the same stirring rate, PET bottle fragments
of the same specifications and particle size were reacted
at a temperature of 200 ℃ for 2 hours, with a feeding
amount of 40 g of ethylene glycol, 10 g of waste PET
bottles, and 0.4 g of catalyst. Three repeated experiments
Figure 3 Yield of PHET at different reaction temperatures were conducted, and the yields of the depolymerized
product BHET were measured to be 84%, 83% and 84%,
3.3 Effect of catalyst dosage on yield respectively.
Under the conditions of a 4:1 mass ratio of ethylene 3.5 Infrared spectra of products and
glycol to PET, a reaction time of 2 hours, and a reaction catalysts
temperature of 200 ℃, PET bottle fragments of the
same specification and particle size were mixed at the
same stirring rate by varying the mass ratio of different
catalysts to reaction materials. The results of measuring
the yield of the reactants are shown in Figure 4. When the
catalyst dosage is low, the concentration of the catalyst
in the reaction system is low, and the catalytic effect is
not significant. PET depolymerization is incomplete, and
non depolymerized PET residue can be found through
filtration, resulting in a low yield of BHET.As the mass
ratio of catalyst to reactants increases, significant PET
Figure 5 Infrared spectrum of BHET depolymerization
depolymerization can be observed, but as the mass ratio of product
catalyst to reactants increases, the measured yield of BHET
does not show a significant increase.However, it resulted The compressed sample spectrum was measured
in catalyst waste and impurities in the depolymerization using a Fourier transform infrared spectrometer TENSOR
product BHET. Therefore, the optimal ratio of catalyst II from Bruker GmbH in Germany, as shown in the figure.Figure
usage to reactants was controlled at 8 ‰. 5 shows the infrared spectrum of the depolymerized product
-1
3.4 Repeated experiments under optimal PHET.The vibration absorption peak at 3280.51 cm in the
process conditions spectrum is the—OH stretching vibration.The stretching
Vol.51,2025 ·3·